December 3rd, 2020
Part One: Inefficiencies and Opportunities in Patient Recruitment and Enrollment
By Kent Siri, VP of Marketing at Reify Health

Clinical research is a cornerstone of our global healthcare ecosystem. More than ever, the public relies on advances in research to fuel therapies for diseases, both new and old. And — as the current pandemic has highlighted — when it comes to clinical trials, speed matters.
Fortunately, the industry has made some significant strides in recent years. To name just a few: the FDA has been more efficient at reviewing and approving drugs, digitization and availability of real-world evidence show significant promise in personalized medicine, and investments in technology have fostered a more patient-centric approach to designing and running clinical trials.
However, we still need to see major gains in overall clinical trial efficiency if we want to reduce costs and shorten clinical development timelines. Despite all the funds poured into clinical trial innovation, the largest source of clinical development delays – patient recruitment and enrollment – continues to stall the development of potentially effective new therapies.
You’ve likely heard some of the statistics:
- 80% of all clinical trials fail to meet their patient enrollment objectives
- 48% of research sites enroll one or fewer patients for a given trial
- Actual enrollment timelines are nearly twice as long as planned timelines
Our industry has spent decades and many billions of dollars developing solutions to do something about these eye-popping statistics. Pharmaceutical sponsors have invested more in patient recruitment strategies that cast wider nets or leverage social, behavioral, and demographic data to target more patients; there are a number of companies that utilize increasingly sophisticated algorithms to engage and refer potential patients to sites; and several companies are beginning to leverage EHRs and AI to identify potential patients already connected to research sites. These solutions have been effective in solving parts of the problem, but we as an industry still have work to do to enroll the majority of trials rapidly and predictably.
To do that work, we need to start focusing on a piece of the puzzle that is just as critical as (and maybe more critical than) patient identification, but has been historically ignored.
When you take a look at the entire patient recruitment and patient enrollment funnel, several key activities – from patient identification, to intake, to pre-screening and screening – rely on research sites and research coordinators to carry them out. Yet these sites, and the research staff therein, have not been given the proper tools or technology to manage and track recruitment efficiently. In fact, our research shows that nearly 90% of clinical research sites still rely on paper and / or spreadsheets to manage this process. Only when patients approach randomization and enrollment do sites have access to tools and tech that aid in process management and data capture. (see fig. 1)
Fig. 1: Site activities across the recruitment and patient enrollment funnel
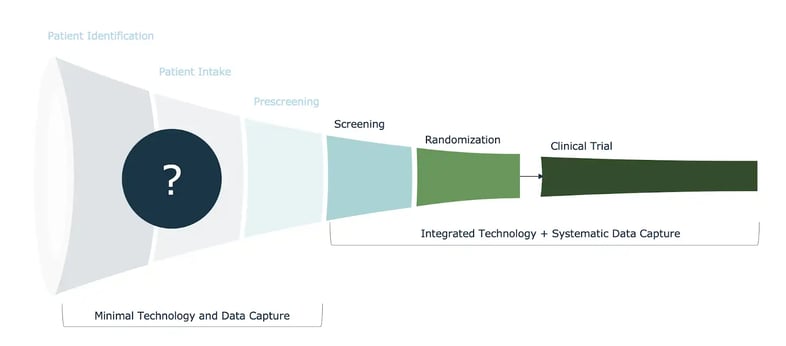
This asymmetry results in gaps in transparency, lost data, and – ultimately – a massive blind spot in clinical research: what’s happening with patients prior to randomization? This is exactly the type of problem we at OneStudyTeam are beginning to solve – we are starting to shine a light on this blind spot. In doing so, we have unveiled some surprising insights and some encouraging opportunities across patient recruitment and enrollment.
The vast majority of potential candidates considered for clinical trials (well over 90 percent) never make it to enrollment. While many of those patients who fall out do so for legitimate reasons (e.g. they have a comorbidity or drug allergy that excludes them from participating), our data show that a meaningful percentage of them could effectively enroll in studies, but are lost due to inefficiencies in how we manage trial operations. In other words, there is a significant amount of “avoidable loss” throughout patient recruitment and enrollment that, if addressed, can have a substantial impact on the success of trials.
If we, as an industry, can better understand where patients are falling out and why, we can meaningfully improve enrollment efficiency and truly improve the speed of clinical trials.
In this blog series, we will focus on 3 key phases of the patient recruitment and patient enrollment funnel. For each, we will begin to quantify the magnitude of attrition and explain key reasons why patients do not progress in the enrollment process:
Potential Patients → Pre-Screened Patients
- We will investigate where our industry identifies potential patients, how sites are managing potential patients across those various channels, and challenges sites face in engaging patients at the top of the patient recruitment funnel.
Pre-Screened Patients → Patients Who Schedule a First Screening Visit
- We will look at the methods sites use to pre-screen patients, types of questions asked during pre-screening, and major causes for pre-screen failures.
Screened Patients → Enrolled / Randomized Patients
- We will dig into why we see drop off between patients who have been successfully screened and patients who actually make it to enrollment.
We aim to use this blog series to create a more comprehensive understanding of the challenges our industry faces at different stages of the patient enrollment process. More importantly, we hope to shed some light on what we as members of the clinical trial ecosystem can do to ensure that more patients have an opportunity to participate in clinical trials and — ultimately — a chance at a better life.
Learn more about solving clinical trial enrollment challenges in our new ebook, Strategies to Fill Gaps in Patient Recruitment and Enrollment.
Related Posts

How Does a Trial Manager in Greece Improve Clinical Trial Operations with StudyTeam®?
Dimitris Tziogas, local trial manager at a biotechnology company in ...
Read More
How to Address Key Clinical Trial Challenges, According to Clinresco Centres in South Africa
There’s no single solution to overcoming a research site’s specific ...
Read More
3 Clinical Trial Billing Challenges Research Sites Solve with StudyTeam
Challenge 1: Complicated coverage analysis Challenge 2: Tedious budgeting ...
Read More
.png?width=64&name=OST%20Transparent%20(1).png)